Light and electrons - Chemical Ideas 6.1 and 2.3
Energy levels
There are verious energy levels that are fixed that an electron can occupy. When the electron is at its closest to the nucleus they are in the ground state at this point they have the lowest possible energy they can have. The difference in energy level decrease the further out the electron moves from the nucleus. This means that when an electron moves up a shell the distance from the shell it is currently on to the next shell is shorter than the distance from the shell below the current to the current shell is less. Electrons cant exist inbetween electron shells instead when they move up a shell they move from the current shell to the next shell instantly.
Absorption spectra
The absorbtion spectrum as seen on earth is the visible light (this is a veriaty of colours that change as you move across the spectra) . The absorbion spectra is used in order to see what element is present in a reaction when light is absorbed in the reaction and as a result cant be seen on the spectra, The elements can be identifed by seing where black lines appear on the spectra.
There are verious energy levels that are fixed that an electron can occupy. When the electron is at its closest to the nucleus they are in the ground state at this point they have the lowest possible energy they can have. The difference in energy level decrease the further out the electron moves from the nucleus. This means that when an electron moves up a shell the distance from the shell it is currently on to the next shell is shorter than the distance from the shell below the current to the current shell is less. Electrons cant exist inbetween electron shells instead when they move up a shell they move from the current shell to the next shell instantly.
Absorption spectra
The absorbtion spectrum as seen on earth is the visible light (this is a veriaty of colours that change as you move across the spectra) . The absorbion spectra is used in order to see what element is present in a reaction when light is absorbed in the reaction and as a result cant be seen on the spectra, The elements can be identifed by seing where black lines appear on the spectra.
The process that takes place when this happens is:
- Electrons absorb photon (package of energy)
- Increase in energy allows electrons to move up to a higher energy level ( this is what causes lines on the absorbsion spectra)
- The more radiation absorbed by the electron the higher the frequency it will have and will move up more shells (the higher the frequency the further to the right the black lines will appear on the spectra)
Emission spectra
The emission spectra is different to the absorbsion spectra as instead of having a coloured background it has a black background and instead of black lines appearing coloured lines appear. This is the energy that is being emitted by the electron as it moves down a shell.
The emission spectra is different to the absorbsion spectra as instead of having a coloured background it has a black background and instead of black lines appearing coloured lines appear. This is the energy that is being emitted by the electron as it moves down a shell.
An emmison spectrum can been seen in a reaction when a chemical is burned with a coloured flame.
- Electrons absorb photon (package of energy)
- Increase in energy allows electrons to move up to a higher energy level
- Electrons drop down to the lower energy levels (This produces the coloured lines on the emisson spectra )
- The radiation emitted from the electrons has a specific frequency depending on wich shells then fall from and which shells that are on after fall
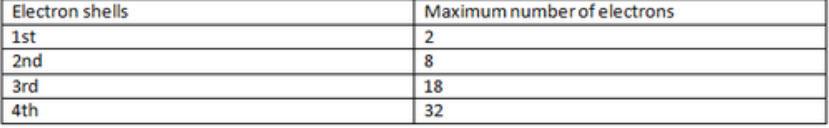
Electron shells
Energy levels is another term for electron shells, each shell can only hold a certain number of electrons before electrons have to be on another outer shell. A periodic table can easily be used in order to find the number of electrons in an atom of an element (see a simple model of the atom)
Energy levels is another term for electron shells, each shell can only hold a certain number of electrons before electrons have to be on another outer shell. A periodic table can easily be used in order to find the number of electrons in an atom of an element (see a simple model of the atom)