Alkanes and Other Hydrocarbons
Modelling and Naming Alkanes
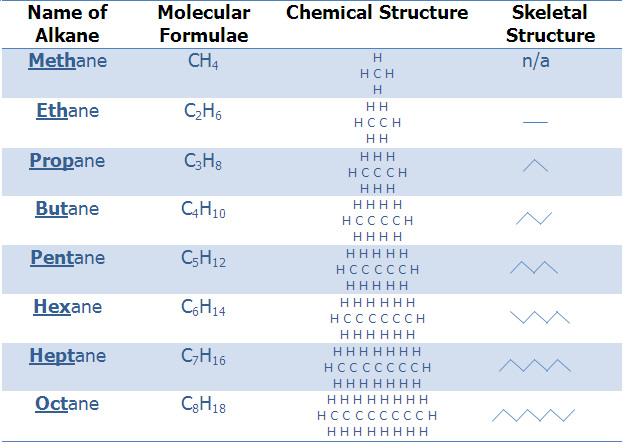
Alkanes are a form of hydrocarbons and therefore are constructed by only hydrogen and carbon atoms. Alkanes follow a specific rule where the number of hydrogen atoms is equal to 2n+2, with n being the number of carbons. Knowing this rule means that a large amount of alkanes can be drawn and sketched in many different ways which are shown in the table below.
Alkanes are a form of hydrocarbons and therefore are constructed by only hydrogen and carbon atoms. Alkanes follow a specific rule where the number of hydrogen atoms is equal to 2n+2, with n being the number of carbons. Knowing this rule means that a large amount of alkanes can be drawn and sketched in many different ways which are shown in the table below.
In the table above the names of the first eight alkanes are given. They all end in –ane therefore are distinguished between there suffix. The first four suffixes (methane, ethane, propane, butane) do not follow a rule of such, but the following alkanes are given the numerical name due to the number of carbons in each. For example hexane is given its suffix because it has six carbons and hex- is the term given to six.
Also in the table is the molecular formula which follows the rule 2n+2, alongside the chemical structure and skeletal structure. These are just two of the ways of representing alkanes. In Alkenes the 2n+2 rule changes to 2n because they include a double bond between two carbons and two less hydrogen atoms are used (eg. C3H6)
The skeletal structure represents only the amount of carbons. Each turn/angle in the line represents a carbon and it is assumed that there are hydrogen atoms in the bonds that are not shown. Also the skeletal structure takes up this shape because the carbon atoms are attempting to repel one another and therefore take up this shape, in which the molecule is most stable. (See below)
Summary – Open Chain alkanes
· Open-chain alkenes follow the general formula CnH2n+2.
· Names end in –ane
· Saturated - all the bonds between the carbon atoms are single
· Aliphatic – they have no ring structures
Other Hydrocarbons - Cycloalkanes
· General formula is CnH2n
· Names end in –ane
· Saturated - all the bonds between the carbon atoms are single
· Not aromatic – they have no benzene structures
Other Hydrocarbons – Alkenes
· General formula is CnH2n
· Names end in –ene
· Unsaturated - all the bonds between the carbon atoms are single
· Aliphatic – they have no benzene structures
Other Hydrocarbons – Arenes
· Names end in –ene
· Unsaturated
· Are aromatic – they have one or more benzene rings in their structres
· Open-chain alkenes follow the general formula CnH2n+2.
· Names end in –ane
· Saturated - all the bonds between the carbon atoms are single
· Aliphatic – they have no ring structures
Other Hydrocarbons - Cycloalkanes
· General formula is CnH2n
· Names end in –ane
· Saturated - all the bonds between the carbon atoms are single
· Not aromatic – they have no benzene structures
Other Hydrocarbons – Alkenes
· General formula is CnH2n
· Names end in –ene
· Unsaturated - all the bonds between the carbon atoms are single
· Aliphatic – they have no benzene structures
Other Hydrocarbons – Arenes
· Names end in –ene
· Unsaturated
· Are aromatic – they have one or more benzene rings in their structres