Chemical bonding and properties - Chemical Ideas 3.1
What type of bond
The type of bond that is formed in a reaction depends on the two atoms that formed it. The factor that has to be taken into account when looking at the type of bonds formed is if its a metal or non metal atoms that are bonding together.
The type of bond that is formed in a reaction depends on the two atoms that formed it. The factor that has to be taken into account when looking at the type of bonds formed is if its a metal or non metal atoms that are bonding together.
Properties depenfing on bonding
Compounds with similiar bonds all have similiar shapes this is caused by the type of bonds that are formed when the compound was created.
Compounds with similiar bonds all have similiar shapes this is caused by the type of bonds that are formed when the compound was created.
Ionic bonding
In nature atoms are usually more stable when they have a full outer shell of electrons. In ionic bonding both elements have a charge as the metal atom transfers a number of electrons to the non-metal electrons this means that the metal now has a positive charge as it lost an electron however the non metal has a negative charge as it has gained an electron. As a result of the atoms gaining and loseing electrons they have a full outer shell. In a ionic lattice atoms atoms arrange themselves so that a positively charged ion is allways next to a negatively charged ion. Positively charged ions are called Cations and negatively charged ions are called Anions. An ionic lattice is held together by electostatic attraction between the Cations and Anions
In nature atoms are usually more stable when they have a full outer shell of electrons. In ionic bonding both elements have a charge as the metal atom transfers a number of electrons to the non-metal electrons this means that the metal now has a positive charge as it lost an electron however the non metal has a negative charge as it has gained an electron. As a result of the atoms gaining and loseing electrons they have a full outer shell. In a ionic lattice atoms atoms arrange themselves so that a positively charged ion is allways next to a negatively charged ion. Positively charged ions are called Cations and negatively charged ions are called Anions. An ionic lattice is held together by electostatic attraction between the Cations and Anions
Covalent bonding
In order to achieve a full outer shell of electrons in covalent bonding atoms will share electrons so that they have full outer shells. In some molecules instead of shareing one electron each to form a pair, a atom will share two electrons with the other atom this is called a dative covalent bond. Covalent bonds are also held together by electrostatic attraction.
In order to achieve a full outer shell of electrons in covalent bonding atoms will share electrons so that they have full outer shells. In some molecules instead of shareing one electron each to form a pair, a atom will share two electrons with the other atom this is called a dative covalent bond. Covalent bonds are also held together by electrostatic attraction.
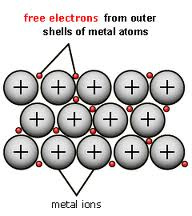
Metallic bonding
In metallic bonding atoms form a similiar shape to a ionic lattice but instead of having positive and negatively charged ions all of the ions are positively charged and the outer shell of electrons are all shared. When electrons can more freely like this they are said to be delocalised. As the electrons can move arround freely they can also conduct electricity better. Metallic bonds are held together by the electrostatic attraction between thee sea of electrons and the positively charged ions.
In metallic bonding atoms form a similiar shape to a ionic lattice but instead of having positive and negatively charged ions all of the ions are positively charged and the outer shell of electrons are all shared. When electrons can more freely like this they are said to be delocalised. As the electrons can move arround freely they can also conduct electricity better. Metallic bonds are held together by the electrostatic attraction between thee sea of electrons and the positively charged ions.