Bond Enthalpies And Calculations - Chemical Ideas 4.2
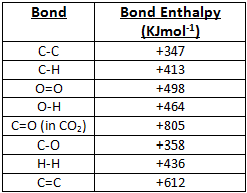
Bond enthalpy is the term used to describe the amount of energy needed to break the bonds in 1 mole of compounds. The values in the table to the left are average values so therefore the true value could slightly vary depending on the values used. For the calculations below we have used the values in the table to the right. Calculating Bond Enthalpies can be broken into the following steps:
- Write a balanced equation for the reaction in the question
- Draw the full structure of all the moleculed involved
- Using the structural diagram from step 2 write out the amounts of each bonds that are to be broken and made.
- Substitue in the values from the table on the right and calculate the energy total.
Heres an example reaction where we follow our step by step guide to
calculate the energy needed to break and make the bonds of the molecules.
Question: Use the bond enthalpy table to calculate the enthalpy of the
combustion of pentane (C5H12) ?
calculate the energy needed to break and make the bonds of the molecules.
Question: Use the bond enthalpy table to calculate the enthalpy of the
combustion of pentane (C5H12) ?
Bond Strengths
- The greater the bond enthalpy, the stronger the bond.
- Short bonds are stronger than long bonds
- C=C is shorter than C-C and therefore has a greater value
Questions
1) Use the bond enthalpy table to calculate the complete combustion of hydrogen?
2) Use the bond enthalpy table to calculate the complete combustion of hexane (C6H14) ?
3) Use the bond enthalpy table to calculate the complete combustion of hept-1-ene
2) Use the bond enthalpy table to calculate the complete combustion of hexane (C6H14) ?
3) Use the bond enthalpy table to calculate the complete combustion of hept-1-ene

